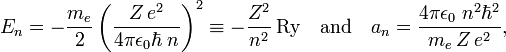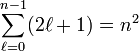Principal quantum number
The principal quantum number, usually designated by n, appears in the description of the electronic structure of atoms. The quantum number first arose in the Bohr-Sommerfeld theory of the hydrogen atom, but it is also part of the solution of the Schrödinger equation for hydrogen-like atoms. It is a positive integral number, n = 1, 2, 3, …, that indexes atomic shells. Historically, atomic shells were indicated by the capital letters K, L, M, … for n=1,2,3,…, respectively, but this usage is dying out.
In the Bohr-Sommerfeld ("old") quantum theory, the electron in a hydrogen-like (one-electron) atom moves in elliptic orbits. The principal quantum number appears in this theory at two places: in the energy En of the electron and in the length an of the major semiaxis of the nth orbit,
where Ry is the Rydberg energy for infinite nuclear mass (= 13.605 6923 eV). Further, me is the mass of the electron, −e is the charge of the electron, Ze is the charge of the nucleus, ε0 is the electric constant, and 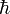 is Planck's reduced constant.
is Planck's reduced constant.
In the "new" quantum mechanics (of Heisenberg, Schrödinger, and others) the energy En of a bound electron in a hydrogen-like atom satisfies the exact same equation, but the electron orbit is replaced by an electron orbital; the latter has no radius. However, in the new quantum theory the same expression for an appears in the form of the expectation value of r (the length of the position vector of the electron) with respect to a state with principal quantum number n. That is, quantum mechanics gives the same measure for the "size" of a one-electron atom (in state n) as the old quantum theory.
Strictly speaking, the principal quantum number is not defined for many-electron atoms. However, in a fairly good approximate description (central field plus independent-particle model) of the many-electron atom, the principal quantum number does appear and hence n is a label that is often applied to many-electron atoms as well.
[edit] Azimuthal and magnetic quantum numbers
An atomic shell consists of atomic subshells that are labeled by the azimuthal quantum number, commonly denoted by ℓ. The azimuthal quantum number is more often referred to as angular momentum quantum number, because the eigenvalues of the squared orbital angular momentum operator 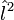 are equal to ℓ(ℓ+1) ℏ².
are equal to ℓ(ℓ+1) ℏ².
For a given atomic shell of principal quantum number n, ℓ runs from 0 to n−1, as follows from the solution of the Schrödinger equation. In total, an atomic shell with quantum number n consists of n subshells and has
spatial (i.e., function of the position vector of the electron) atomic orbitals.
An atomic subshell consists of 2ℓ+1 atomic orbitals labeled by the orbital magnetic quantum number, almost invariably denoted by m. For given ℓ, m runs over 2ℓ+1 values: m = −ℓ, −ℓ+1, …, ℓ−1, ℓ. The number m is proportional to the eigenvalues of the z-component of the orbital angular momentum operator 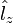 that has eigenvalues mℏ.
that has eigenvalues mℏ.
For historical reasons the orbitals of certain azimuthal quantum number ℓ are denoted by letters: s, p, d, f, g, for ℓ = 0, 1, 2, 3, 4, respectively. For instance an atomic orbital with n = 4 and ℓ = 2 is written as 4d. If the n = 4, ℓ = 2 subshell is occupied k times (there are k electrons in the 4d orbital), we indicate this by writing (4d)k.
Any spatial atomic orbital can be occupied at most twice, which is because the spin magnetic quantum number ms (proportional to the eigenvalues of the z-component of the spin angular momentum operator 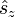 ) can have only two values: +½ (spin up) and −½ (spin down). In addition, the Pauli exclusion principle states that no two electrons with the same four quantum numbers n, ℓ, m, and ms can occupy the same atomic orbital. As a consequence, the spatial orbital (n,ℓ,m) can be occupied at most by two electrons with spin ms = ±½.
Hence a subshell can accommodate at most 2(2l+1) electrons. If a subshell accommodates the maximum number of electrons, it is called closed. If the atomic shell n contains the maximum of 2n2 electrons, it is also called closed. For instance the noble gas neon in its lowest energy state has the electron configuration (1s)2(2s)2(2p)6, that is, all its shells and subshells are closed.
) can have only two values: +½ (spin up) and −½ (spin down). In addition, the Pauli exclusion principle states that no two electrons with the same four quantum numbers n, ℓ, m, and ms can occupy the same atomic orbital. As a consequence, the spatial orbital (n,ℓ,m) can be occupied at most by two electrons with spin ms = ±½.
Hence a subshell can accommodate at most 2(2l+1) electrons. If a subshell accommodates the maximum number of electrons, it is called closed. If the atomic shell n contains the maximum of 2n2 electrons, it is also called closed. For instance the noble gas neon in its lowest energy state has the electron configuration (1s)2(2s)2(2p)6, that is, all its shells and subshells are closed.
[edit] Bibliography
- L. Pauling and E. B. Wilson, Introduction to Quantum Mechanics, McGraw-Hill, New York (1935).
- J. C. Slater, Quantum Theory of Atomic Structure, vol. I, McGraw-Hill, New York (1960).