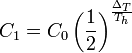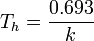Half-life
For any reactant subject to first-order decomposition, the amount of time needed for one half of the substance to decay is referred to as the half-life of that compound. Although the term is often associated with radioactive decay, it also applies equally to chemical decomposition, such as the decomposition of azomethane (CH3N=NCH3) into methane and nitrogen gas. Many compounds decay so slowly that it is impractical to wait for half of the material to decay to determine the half-life. In such cases, a convenient fact is that the half-life is 693 times the amount of time required for 0.1% of the substance to decay. Using the value of the half-life of a compound, one can predict both future and past quantities.
[edit] Mathematics
The future concentration of a substance, C1, after some passage of time ΔT, can easily be calculated if the present concentration, C0, and the half-life, Th, are known:
For a reaction is the first-order for a particular reactant, A, and first-order overall, the chemical rate constant for the reaction, k, is related to the half-life Th by this equation:
| |
Some content on this page may previously have appeared on Citizendium. |